Imaging progression
Imaging is increasingly valuable in detecting progression in early glaucoma
Recent advances in imaging technology have given clinicians valuable new tools for tracking glaucoma progression over time and fine-tuning therapy in the early stages of glaucomatous disease, according to Francesco Oddone MD, PhD.
“Imaging is expected to be more sensitive than visual field in predicting conversion and detecting progression, especially in early glaucoma. At the moment, however, there is no consensus on the best technique or criteria to detect glaucoma conversion or progression, or to assess what amount of change would be clinically meaningful,” he told delegates attending Glaucoma Day as part of the XXXV Congress of the ESCRS in Lisbon.
Dr Oddone, Head of the Glaucoma Unit at Britannic Hospital, Rome, Italy, noted that macular parameters seem to be more consistent than retinal nerve fibre layer (RNFL) parameters in detecting progression across the entire glaucoma severity spectrum, but might also be more affected by ageing.
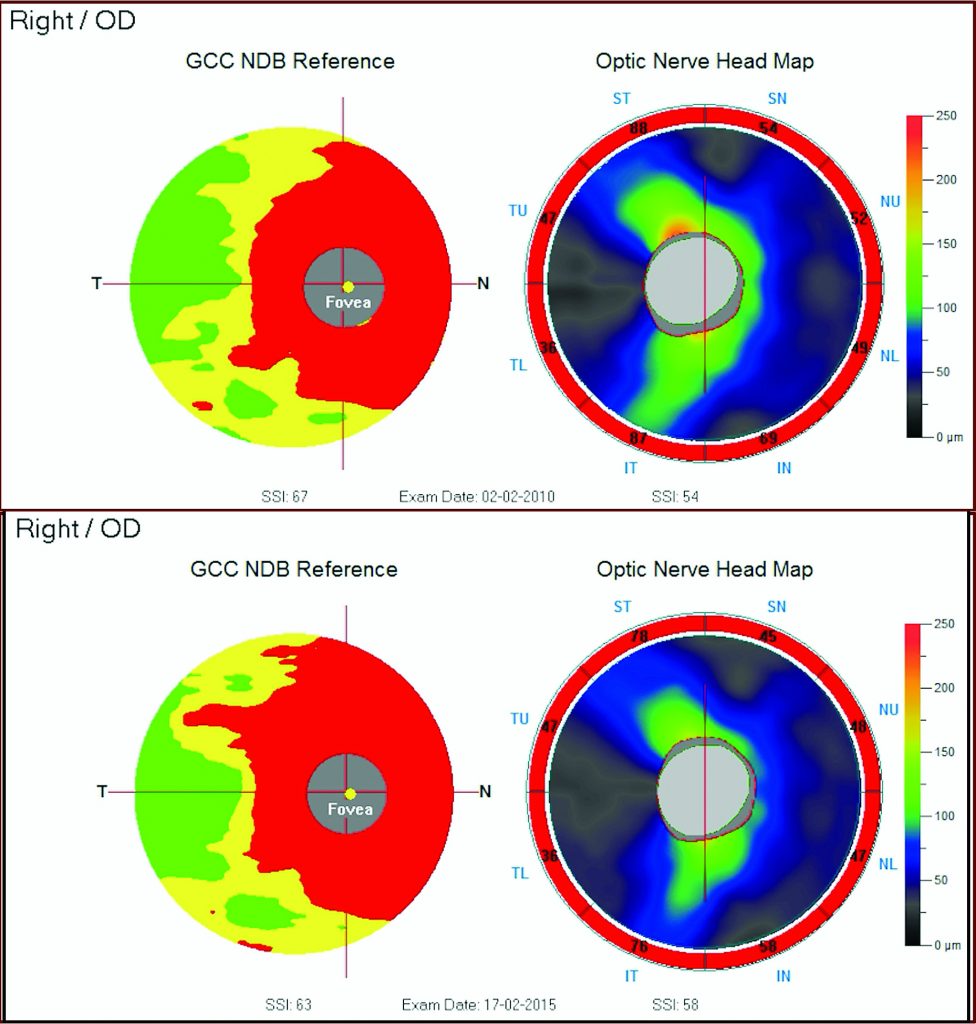 Monitoring with imaging - 2006-2011
Monitoring with imaging - 2006-2011
Given the uncertainty in interpreting the results of imaging scans and the cost to perform each examination, clinicians should appraise which tests provide the most useful information for their specific decision, he advised.
“Examinations whose results are unlikely to influence clinical decisions should be replaced with more frequent examinations or tests that do influence clinical decisions. Approaches that combine structural and functional measurements will likely improve the detection of glaucoma progression while correlating more with patients’ relevant outcomes,” he said.
The goal of glaucoma treatment is to slow down or halt the progression of the disease in order to maintain the patient’s quality of life at a sustainable cost, said Dr Oddone.
“Assessing the progression of the disease reliably is thus critical for decision-making in glaucoma. We need to know when to start treatment, change an ongoing medical treatment, switch medical treatment to surgical interventions and assess the individual risk of visual disability. These actions may have consequences on a patient’s well-being and quality of life and must be based on solid clinical information,” he said.
Anatomical changes in glaucoma are either preceded, accompanied or followed by clinically detectable functional changes in the form of progressive visual field defects, said Dr Oddone.
“Only functional changes are currently considered as relevant endpoints of glaucoma care both by clinicians and by regulatory authorities. Visual field can in fact be considered a validated surrogate endpoint of quality of life measurements, which are less practical to be measured clinically in a consistent and reliable manner,” he said.
The rationale for using structural endpoints derived from OCT images is that they may be more consistent than psycophysical endpoints and are not affected by a learning curve.
“But considering the extra costs involved, imaging is expected to provide information that is relevant for glaucoma management in a clinically meaningful way, in addition to the information provided by psycophysical testing,” he said.
Imaging is expected to provide information that is relevant for glaucoma management
“Nevertheless, regulatory authorities, such as the US FDA, for validation of structural endpoints, request such a strong correlation (R2 of 0.9) to current vision or future vision loss that structural endpoints would never be validated if these criteria will be not revised,” said Dr Oddone.
The challenge for the physician, however, is knowing how much of a structural change is clinically significant or what change in vision would be expected to occur, and when, as a result of this structural change, said Dr Oddone.
Some recent studies have sought to shed some light on a few of these questions with some measure of success, he said. Zhang et al., for instance, took VF measurements and OCT scans in 417 glaucoma suspect/pre-perimetric glaucoma eyes and 377 perimetric glaucoma eyes every six months for several years.
“They showed that baseline reductions in NFL and ganglion cell complex (GCC) thickness can predict the development of VF loss in glaucoma suspects and pre-perimetric glaucoma. Eyes with a borderline or abnormal OCT finding had a five-time greater chance of conversion. Focal GCC and RNFL loss were also found to be the strongest predictors for VF progression in perimetric glaucoma,” he said.
Francesco Oddone: oddonef@gmail.com
Authors
Dermot McGrath
Published
Wednesday, January 30, 2019
Category